Complete volume with articles 1 to 35 - Cucurbit Breeding - North ...
Complete volume with articles 1 to 35 - Cucurbit Breeding - North ... Complete volume with articles 1 to 35 - Cucurbit Breeding - North ...
Table 1. Influence of light and culture conditions on Cucumis metuliferus protoplasts division. z Culture Light Day 4 Day 8 conditions (!mol.m -2.s -1 ) Chamber PD4-1 y PD4-2 x PD8-1 y PD8-2 x Agarose 13.5 1 27 3.1 25 29 - 2 27 2.2 37 21 Liquid 13.5 1 7 0.0 17 3 - 2 7 0.1 12 2 LSD (5%) 4 1.3 6 6 CV (%) 13 51 17 27 z,y Percentages of protoplasts which divided once and more than once. Table 2. Protoplast isolation yields of different genotype and tissues. z Tissue Protoplast Protoplast Genotype source yield (x106/g)y viability (%) Cucumis metuliferus PI 482452 Cotyledon 4.1 ± 1.5x 66.3 ± 30.6x PI 482461 Cotyledon 7.9 ± 0.4 73.4 ± 17.0 PI 482454 Cotyledon 7.5 ± 2.3 77.9 ± 16.3 PI 482454 Leaf 13.0 ± 2.0 69.2 ± 8.4 Cucumis sativus Sumter Cotyledon 2.4 ± 0.5 63.7 ± 19.1 Wis. SMR 18 Cotyledon 9.1 ± 0.5 78.6 ± 8.1 Wis. SMR 18 Leaf 3.5 ± 1.1 62.3 ± 8.9 z Data are means of 3 replications. y Viable protoplasts released/g fresh weight of material. Table 3. Effect of center disc size and low protoplast densities on the percent multiple protoplast division 4 days after isolation (%). z Protoplast density (no./ml agarose) Disc size 5x102 1x103 1x104 10 !l 0.4 1.1 3.9 50 !l 0.4 3.0 4.2 z Data are means of 7 combinations of genotypes and tissue source (see table 2). Each combination has 3 replications, 3 samples and 2 subsamples. Cucurbit Genetics Cooperative Report 24:97-101 (2001) 99
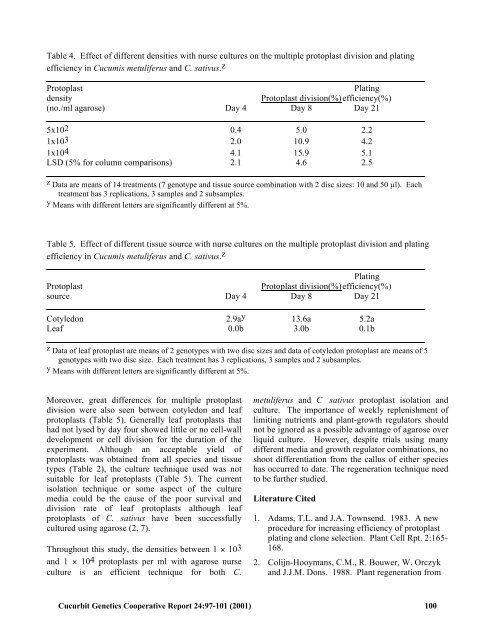
Table 4. Effect of different densities with nurse cultures on the multiple protoplast division and plating efficiency in Cucumis metuliferus and C. sativus. z Protoplast Plating density Protoplast division(%) efficiency(%) (no./ml agarose) Day 4 Day 8 Day 21 5x102 0.4 5.0 2.2 1x103 2.0 10.9 4.2 1x104 4.1 15.9 5.1 LSD (5% for column comparisons) 2.1 4.6 2.5 z Data are means of 14 treatments (7 genotype and tissue source combination with 2 disc sizes: 10 and 50 !l). Each treatment has 3 replications, 3 samples and 2 subsamples. y Means with different letters are significantly different at 5%. Table 5. Effect of different tissue source with nurse cultures on the multiple protoplast division and plating efficiency in Cucumis metuliferus and C. sativus. z Plating Protoplast Protoplast division(%) efficiency(%) source Day 4 Day 8 Day 21 Cotyledon 2.9a y 13.6a 5.2a Leaf 0.0b 3.0b 0.1b z Data of leaf protoplast are means of 2 genotypes with two disc sizes and data of cotyledon protoplast are means of 5 genotypes with two disc size. Each treatment has 3 replications, 3 samples and 2 subsamples. y Means with different letters are significantly different at 5%. Moreover, great differences for multiple protoplast division were also seen between cotyledon and leaf protoplasts (Table 5). Generally leaf protoplasts that had not lysed by day four showed little or no cell-wall development or cell division for the duration of the experiment. Although an acceptable yield of protoplasts was obtained from all species and tissue types (Table 2), the culture technique used was not suitable for leaf protoplasts (Table 5). The current isolation technique or some aspect of the culture media could be the cause of the poor survival and division rate of leaf protoplasts although leaf protoplasts of C. sativus have been successfully cultured using agarose (2, 7). Throughout this study, the densities between 1 ! 103 and 1 ! 104 protoplasts per ml with agarose nurse culture is an efficient technique for both C. metuliferus and C sativus protoplast isolation and culture. The importance of weekly replenishment of limiting nutrients and plant-growth regulators should not be ignored as a possible advantage of agarose over liquid culture. However, despite trials using many different media and growth regulator combinations, no shoot differentiation from the callus of either species has occurred to date. The regeneration technique need to be further studied. Literature Cited 1. Adams, T.L. and J.A. Townsend. 1983. A new procedure for increasing efficiency of protoplast plating and clone selection. Plant Cell Rpt. 2:165- 168. 2. Colijn-Hooymans, C.M., R. Bouwer, W. Orczyk and J.J.M. Dons. 1988. Plant regeneration from Cucurbit Genetics Cooperative Report 24:97-101 (2001) 100
- Page 59 and 60: In the present study C. lanatus ent
- Page 61 and 62: Lakes, NJ) was a 0.5 cc (50 unit) s
- Page 63 and 64: Characterization of a New Male Ster
- Page 65 and 66: Table 1. Progeny test of male steri
- Page 67 and 68: Progeny Diploid Year Season generat
- Page 69 and 70: elatively small during its developm
- Page 71 and 72: content had no significant effect o
- Page 73 and 74: Stars-N-Stripes 26.3 12.0 35 8.5 5.
- Page 75 and 76: 11. Mozumder, B.K.G, N.E. Caroselli
- Page 77 and 78: Table 1. Vine length of watermelon
- Page 79 and 80: Survey of Watermelon Trialing Metho
- Page 81 and 82: assigned to adjacent plots (Table 2
- Page 83 and 84: Variation among tropical pumpkin cu
- Page 85 and 86: Table 1. Aggressiveness of powdery
- Page 87 and 88: A large variability of aggressivene
- Page 89 and 90: Table 1: Observed reaction of melon
- Page 91 and 92: Same Gene for Bush Growth Habit in
- Page 93 and 94: Relationship between Fruit Shape an
- Page 95 and 96: Table 3. Seed yield per fruit and s
- Page 97 and 98: The four cultivar-groups differed m
- Page 99 and 100: Lee et al. (17) developed a RAPD ma
- Page 101 and 102: 19. Lelley, T and S. Henglmüller,
- Page 103 and 104: Those primers that had strong bands
- Page 105 and 106: Genetic Variability in Pumpkin (Cuc
- Page 107 and 108: those obtained by the ANOVA and the
- Page 109: Agarose discs were prepared as desc
- Page 113 and 114: Isolation and Callus Production fro
- Page 115 and 116: Table 1. Components used for cultur
- Page 117 and 118: 6. Jia, Shi-rong, You-ying Fu and Y
- Page 119 and 120: the preceding one or two days (depe
- Page 121 and 122: Gene List 2001 for Cucumber Jiahua
- Page 123 and 124: Ar - Anthracnose resistance. One of
- Page 125 and 126: de I determinate habit. Short vine
- Page 127 and 128: gi - ginkgo. Leaves reduced and dis
- Page 129 and 130: Mdh-4 Mdh- 3 Malate dehydrogenase-4
- Page 131 and 132: h from 'Gy 2 cp cp', 'Spartan Salad
- Page 133 and 134: wf - White flesh. Intense white fle
- Page 135 and 136: M16056 Cotyledon cDNA Encoding ribu
- Page 137 and 138: (for CsPK2.2) AB001590 Hypocotyl RN
- Page 139 and 140: Caruth, T. F. 1975. A genetic study
- Page 141 and 142: John, C. A. and J. D. Wilson. 1952.
- Page 143 and 144: Ohkawa, J., A. Shinmyo, M. Kanchana
- Page 145 and 146: Shimizu, S., K. Kanazawa, and A. Ka
- Page 147 and 148: Youngner, V. B. 1952. A study of th
- Page 149 and 150: Cucurbit Genetics Cooperative 2001-
- Page 151 and 152: Denlinger, Phil Mt. Olive Pickle Co
- Page 153 and 154: Keita, Sugiyama Kurume Branch, Natl
- Page 155 and 156: germplasm evaluation and conservati
- Page 157 and 158: Sarfatti, Matti Hazera Ltd., Resear
- Page 159 and 160: Alabama Fenny Dane Arkansas Ted Mor
Table 4. Effect of different densities <strong>with</strong> nurse cultures on the multiple pro<strong>to</strong>plast division and plating<br />
efficiency in Cucumis metuliferus and C. sativus. z<br />
Pro<strong>to</strong>plast Plating<br />
density Pro<strong>to</strong>plast division(%) efficiency(%)<br />
(no./ml agarose) Day 4 Day 8 Day 21<br />
5x102 0.4 5.0 2.2<br />
1x103 2.0 10.9 4.2<br />
1x104 4.1 15.9 5.1<br />
LSD (5% for column comparisons) 2.1 4.6 2.5<br />
z Data are means of 14 treatments (7 genotype and tissue source combination <strong>with</strong> 2 disc sizes: 10 and 50 !l). Each<br />
treatment has 3 replications, 3 samples and 2 subsamples.<br />
y Means <strong>with</strong> different letters are significantly different at 5%.<br />
Table 5. Effect of different tissue source <strong>with</strong> nurse cultures on the multiple pro<strong>to</strong>plast division and plating<br />
efficiency in Cucumis metuliferus and C. sativus. z<br />
Plating<br />
Pro<strong>to</strong>plast Pro<strong>to</strong>plast division(%) efficiency(%)<br />
source Day 4 Day 8 Day 21<br />
Cotyledon 2.9a y 13.6a 5.2a<br />
Leaf 0.0b 3.0b 0.1b<br />
z Data of leaf pro<strong>to</strong>plast are means of 2 genotypes <strong>with</strong> two disc sizes and data of cotyledon pro<strong>to</strong>plast are means of 5<br />
genotypes <strong>with</strong> two disc size. Each treatment has 3 replications, 3 samples and 2 subsamples.<br />
y Means <strong>with</strong> different letters are significantly different at 5%.<br />
Moreover, great differences for multiple pro<strong>to</strong>plast<br />
division were also seen between cotyledon and leaf<br />
pro<strong>to</strong>plasts (Table 5). Generally leaf pro<strong>to</strong>plasts that<br />
had not lysed by day four showed little or no cell-wall<br />
development or cell division for the duration of the<br />
experiment. Although an acceptable yield of<br />
pro<strong>to</strong>plasts was obtained from all species and tissue<br />
types (Table 2), the culture technique used was not<br />
suitable for leaf pro<strong>to</strong>plasts (Table 5). The current<br />
isolation technique or some aspect of the culture<br />
media could be the cause of the poor survival and<br />
division rate of leaf pro<strong>to</strong>plasts although leaf<br />
pro<strong>to</strong>plasts of C. sativus have been successfully<br />
cultured using agarose (2, 7).<br />
Throughout this study, the densities between 1 ! 103<br />
and 1 ! 104 pro<strong>to</strong>plasts per ml <strong>with</strong> agarose nurse<br />
culture is an efficient technique for both C.<br />
metuliferus and C sativus pro<strong>to</strong>plast isolation and<br />
culture. The importance of weekly replenishment of<br />
limiting nutrients and plant-growth regula<strong>to</strong>rs should<br />
not be ignored as a possible advantage of agarose over<br />
liquid culture. However, despite trials using many<br />
different media and growth regula<strong>to</strong>r combinations, no<br />
shoot differentiation from the callus of either species<br />
has occurred <strong>to</strong> date. The regeneration technique need<br />
<strong>to</strong> be further studied.<br />
Literature Cited<br />
1. Adams, T.L. and J.A. Townsend. 1983. A new<br />
procedure for increasing efficiency of pro<strong>to</strong>plast<br />
plating and clone selection. Plant Cell Rpt. 2:165-<br />
168.<br />
2. Colijn-Hooymans, C.M., R. Bouwer, W. Orczyk<br />
and J.J.M. Dons. 1988. Plant regeneration from<br />
<strong>Cucurbit</strong> Genetics Cooperative Report 24:97-101 (2001) 100