mmpc - National Indian Health Board
mmpc - National Indian Health Board
mmpc - National Indian Health Board
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
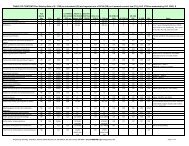
RRIAR<br />
Ref.<br />
#<br />
20.<br />
23.<br />
26.<br />
28.a.<br />
Short Title/<br />
Current Status of Regulation/<br />
Title/<br />
Agency<br />
Assuring Access to Services<br />
ACTION: Proposed Rule<br />
NOTICE: Medicaid Program;<br />
Methods for Assuring Access to<br />
Covered Medicaid Services<br />
AGENCY: CMS<br />
Request for Approval of<br />
Medicaid and CHIP Standard<br />
Forms<br />
ACTION: Request for Comment<br />
NOTICE: Clearance for Medicaid<br />
and CHIP State Plan, Waiver, and<br />
Program Submissions<br />
AGENCY: CMS<br />
Medicaid Home <strong>Health</strong><br />
ACTION: Proposed Rule<br />
NOTICE: Medicaid Program; Faceto-Face<br />
Requirements for Home<br />
<strong>Health</strong> Services; Policy Changes<br />
and Clarifications Related to HH<br />
AGENCY: CMS<br />
Medicaid Eligibility Under ACA<br />
ACTION: Proposed Final Rule<br />
NOTICE: Medicaid; Eligibility<br />
Changes Under the ACA<br />
AGENCY: CMS<br />
TABLE A: REGULATIONS INCLUDED TO DATE IN RRIAR TABLES B AND C<br />
UPDATED THROUGH 11/9/2012<br />
File Code<br />
CMS-2328-P<br />
Dates (Issue, Due, File,<br />
Subsequent Action)<br />
Issue Date: 5/6/2011<br />
Due Date: 7/5/2011<br />
NIHB File Date: 7/5/2011 (filed<br />
by ANHTC)<br />
Date of Subsequent Agency<br />
Action, if any:<br />
CMS-10398 Issue Date: 7/1/2011<br />
Due Date: 8/30/2011<br />
NIHB File Date: None<br />
Date of Subsequent Agency<br />
Action, if any:<br />
CMS-2348-P Issue Date: 7/12/2011<br />
Due Date: 9/12/2011<br />
NIHB File Date: None<br />
Date of Subsequent Agency<br />
Action, if any: Issued Proposed<br />
Rule on home health payment<br />
rates 7/12/2011 (CMS-1353-P)<br />
CMS-2349-PF Issue Date: 8/12/2011<br />
Due Date: 10/31/2011<br />
NIHB File Date: 10/31/2011<br />
Date of Subsequent Agency<br />
Action, if any: Issued Final<br />
Rule 3/23/2012 (see 28.b.)<br />
In Table B--<br />
Is the summary of<br />
Agency action<br />
included?<br />
Is the NIHB analysis<br />
included?<br />
Summary of Agency<br />
action: √<br />
NIHB analysis of<br />
action: √<br />
Summary of Agency<br />
action: √<br />
NIHB analysis of<br />
action: √ (Limited)<br />
Summary of Agency<br />
action: √<br />
NIHB analysis of<br />
action: None.<br />
Summary of Agency<br />
action: √<br />
NIHB analysis of<br />
action: √<br />
In Table C--<br />
Is the list of NIHB<br />
recommendations included?<br />
Has the Agency taken subsequent<br />
action?<br />
Is an analysis of subsequent<br />
Agency action included?<br />
NIHB recommendations included: √<br />
(ANHTC recommendations)<br />
Subsequent Agency action:<br />
Analysis of Agency action:<br />
NIHB recommendations included:<br />
None.<br />
Subsequent Agency action:<br />
Analysis of Agency action:<br />
NIHB recommendations included:<br />
None.<br />
Subsequent Agency action:<br />
Analysis of Agency action:<br />
NIHB recommendations included: √<br />
Subsequent Agency action: √<br />
Analysis of Agency action: To be<br />
completed.<br />
: regulation review complete : regulation currently under review : regulation release pending<br />
<strong>National</strong> <strong>Indian</strong> <strong>Health</strong> <strong>Board</strong>, Regulation Review and Impact Analysis Report Page 4 of 38 11/12/2012