mmpc - National Indian Health Board
mmpc - National Indian Health Board
mmpc - National Indian Health Board
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
RRIAR<br />
Ref.<br />
#<br />
1.c.<br />
8.<br />
9.<br />
13.<br />
Short Title/<br />
Current Status of Regulation/<br />
Title/<br />
Agency<br />
HIT Revised Standards--CMS<br />
EHR Program<br />
ACTION: Proposed Final Rule<br />
NOTICE: <strong>Health</strong> Information Tech.;<br />
Revised Standards, Implementation<br />
Specs and Cert. Criteria for EHRs<br />
AGENCY: ONC, HHS<br />
Sec. 1115 Waiver Transparency<br />
ACTION: Proposed Final Rule<br />
NOTICE: Medicaid; Review/App.<br />
Process for Sec. 1115 Demo’s<br />
AGENCY: CMS<br />
Med/Med Provider Survey<br />
ACTION: Final Rule w/Comments<br />
NOTICE: Medicare, Medicaid, and<br />
CHIP Programs; Additional<br />
Screening Requirements, App. Fee<br />
AGENCY: CMS<br />
Provider Complaint Filing<br />
ACTION: Proposed Rule<br />
NOTICE: Medicare & Medicaid<br />
Providers & Suppliers to notify<br />
beneficiaries of the right to file<br />
written complaint with QIO<br />
AGENCY: CMS<br />
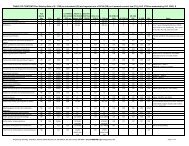
TABLE A: REGULATIONS INCLUDED TO DATE IN RRIAR TABLES B AND C<br />
UPDATED THROUGH 11/9/2012<br />
File Code<br />
HHS<br />
RIN 0991-<br />
AB82<br />
Dates (Issue, Due, File,<br />
Subsequent Action)<br />
Issue Date: 3/7/2012<br />
Due Date: 5:00 pm, 5/7/2012<br />
NIHB File Date: None<br />
Date of Subsequent Agency<br />
Action, if any: Issued Final Rule<br />
9/4/2012<br />
CMS-2325-PF Issue Date: 9/17/2010<br />
Due Date: 11/16/2010<br />
NIHB File Date: 11/15/2010<br />
Date of Subsequent Agency<br />
Action, if any: Issued Final Rule<br />
2/27/2012<br />
CMS-6028-FC Issue Date: 9/23/2010<br />
Due Date: 11/16/2010<br />
NIHB File Date: 11/16/2010<br />
Date of Subsequent Agency<br />
Action, if any: 2/1/2011<br />
Additional: NIHB provided<br />
CMS-3225-P<br />
RIN 0938-<br />
AP94<br />
examples of hardship 3/25/11<br />
Issue Date: 2/2/2011<br />
Due Date: 4/4/2011<br />
NIHB File Date: 4/4/2011<br />
Date of Subsequent Agency<br />
action, if any: Sent Final Rule<br />
to OMB for review 6/2/2011<br />
In Table B--<br />
Is the summary of<br />
Agency action<br />
included?<br />
Is the NIHB analysis<br />
included?<br />
Summary of Agency<br />
action: √<br />
NIHB analysis of<br />
action: None.<br />
Summary of Agency<br />
action: √<br />
Summary of Agency<br />
action: √<br />
TTAG analysis of<br />
action: √<br />
Summary of Agency<br />
action: √<br />
NIHB analysis of<br />
action: √<br />
Summary of Agency<br />
action: √<br />
NIHB analysis of<br />
action: √<br />
In Table C--<br />
Is the list of NIHB<br />
recommendations included?<br />
Has the Agency taken subsequent<br />
action?<br />
Is an analysis of subsequent<br />
Agency action included?<br />
NIHB recommendations included:<br />
Subsequent Agency action:<br />
Analysis of Agency action:<br />
TTAG recommendations included: √<br />
Subsequent Agency action: √<br />
Analysis of Agency action: To be<br />
entered.<br />
NIHB recommendations included: √<br />
Subsequent Agency action: √<br />
Analysis of Agency action: √<br />
NIHB recommendations included: √<br />
Subsequent Agency action: None as<br />
of 5/31/2011.<br />
Analysis of Agency action:<br />
: regulation review complete : regulation currently under review : regulation release pending<br />
<strong>National</strong> <strong>Indian</strong> <strong>Health</strong> <strong>Board</strong>, Regulation Review and Impact Analysis Report Page 2 of 38 11/12/2012