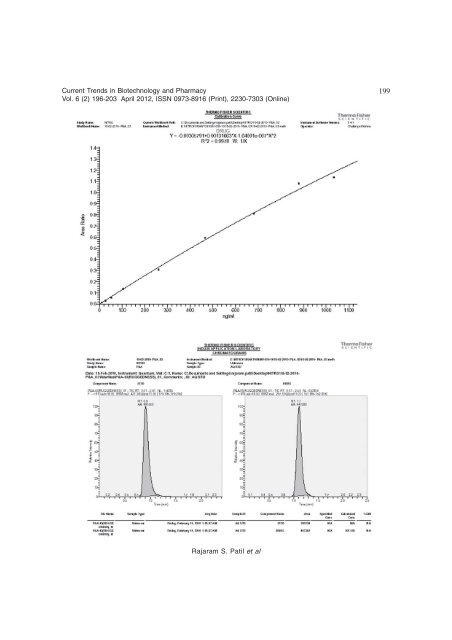
; Current Trends in <strong>Biotechnology</strong> <strong>and</strong> <strong>Pharmacy</strong> Vol. 6 (2) 196-203 April 2012, ISSN 0973-8916 (Print), 2230-7303 (Online) was performed using multiple reaction monitoring (MRM). Results <strong>and</strong> Discussion Development conditions for rapid extraction <strong>of</strong> Nitr<strong>of</strong>urantoin from Human Plasma: Reported extraction techniques for determination <strong>of</strong> nitr<strong>of</strong>urantoin from human plasma [3-6, 8-11, 14-19] is more laborious <strong>and</strong> involves more tedious process like derivatisation. More over, they involve cost efforts <strong>and</strong> in addition to this, they are time consuming. While developing a method, one needs to look at the chromatographic conditions <strong>and</strong> extraction process for minimizing the disadvantages <strong>of</strong> older techniques or methods. In the process, mobile phase selection <strong>and</strong> optimization, column selection is critical to minimize run time, solvent consumption <strong>and</strong> injection volumes. It was observed that, a novel method can be developed with advantages that will eliminate derivatisation, more solvent consumption, high run time <strong>and</strong> rising costs. The new method should <strong>of</strong>fer the benefits <strong>of</strong> using small columns, reducing plasma <strong>and</strong> solvent consumption to arrive at an extraction procedure which will have more extraction recoveries. Conditions for simple <strong>and</strong> rapid HPLC separation with MS/MS Electro-spray Negative ionization mode were developed using an isocratic elution with a mobile phase composed <strong>of</strong> Acetonitrile <strong>and</strong> 5mM Ammonium acetate at a ratio <strong>of</strong> 60:40% v/v. Thus the ions formed for drug <strong>and</strong> Internal st<strong>and</strong>ard in ESI Negative mode due to the addition <strong>of</strong> Hydroxyl ion (OH- ) in carbonyl function (C=O) present in the drug (Fig.1). These conditions gave a well defined, sharp peak <strong>of</strong> Nitr<strong>of</strong>urantoin <strong>and</strong> Losartan (ISTD) with a retention time <strong>of</strong> approximately 1.21 <strong>and</strong> 1.51minutes. Under these conditions an amount <strong>of</strong> Nitr<strong>of</strong>urantoin as low as 1ng/mL could be detected. With these retention times, analysis could be completed in about 3.0 minutes. Method validation Linearity: The quantification <strong>of</strong> the chromatogram was performed using the peak 198 area ratio <strong>of</strong> Nitr<strong>of</strong>urantoin <strong>and</strong> Losartan (ISTD). Nine st<strong>and</strong>ard solutions were prepared. (10.248, 25.621, 51.242, 102.484, 256.209, 457.515, 667.906, 861.814 ng/mL <strong>and</strong> 1013.899 ng/mL) <strong>and</strong> subjected analyses by HPLC-MS/ MS. Three precision <strong>and</strong> accuracy (P&A) batches were injected. The peak area ratio was determined <strong>and</strong> plotted versus the concentration <strong>of</strong> Nitr<strong>of</strong>urantoin. Statistical analysis using least square regression analysis indicated excellent linearity for Nitr<strong>of</strong>urantoin with the concentration range studied as shown in Table 1. In constructing the st<strong>and</strong>ard curve, samples <strong>of</strong> Nitr<strong>of</strong>urantoin in Human Plasma identical to those in the st<strong>and</strong>ard solutions were prepared <strong>and</strong> the Nitr<strong>of</strong>urantoin response ratios were plotted against the concentrations <strong>of</strong> Nitr<strong>of</strong>urantoin in ng/mL as shown in Fig. 2. The linearity <strong>of</strong> the concentration <strong>and</strong> response relation was established over the range <strong>of</strong> 10.248 – 1013.899 ng/mL (R 2 = 0.9898). Fig. 3 shows the LC-MS/MS chromatograms <strong>of</strong> pure drug. (Nitr<strong>of</strong>urantoin), Fig. 4 shows the LC- MS/MS chromatograms <strong>of</strong> drug-free Human plasma <strong>and</strong> Fig. 5 shows the LC-MS/MS chromatograms <strong>of</strong> st<strong>and</strong>ard Plasma sample containing the drug at a concentration <strong>of</strong> 10.248ng/mL. Accuracy <strong>and</strong> precision: The intra-day accuracy <strong>and</strong> precision <strong>of</strong> the assay was evaluated by analyzing six replicates <strong>of</strong> the Plasma containing Nitr<strong>of</strong>urantoin at three different concentrations. The intra-day precisions <strong>of</strong> the analyzed samples are determined by Relative St<strong>and</strong>ard Deviation (RSD) range from 2.11% to 11.01%, while the intra-day accuracy ranged from 83.61% to 107.16%. The inter-day precision <strong>of</strong> the assay was measured by analyzing six replicates <strong>of</strong> Nitr<strong>of</strong>urantoin Plasma samples for three consecutive days. The interday precision <strong>of</strong> the analyzed samples as determined by Relative St<strong>and</strong>ard Deviation (RSD) range from 6.48% to 12.37%, while the inter-day accuracy ranged from 93.13to 103.02%. Nitr<strong>of</strong>urantoin in Human Plasma by using Liquid Chromatography / T<strong>and</strong>em Mass Spectrometry
Current Trends in <strong>Biotechnology</strong> <strong>and</strong> <strong>Pharmacy</strong> Vol. 6 (2) 196-203 April 2012, ISSN 0973-8916 (Print), 2230-7303 (Online) Rajaram S. Patil et al 199