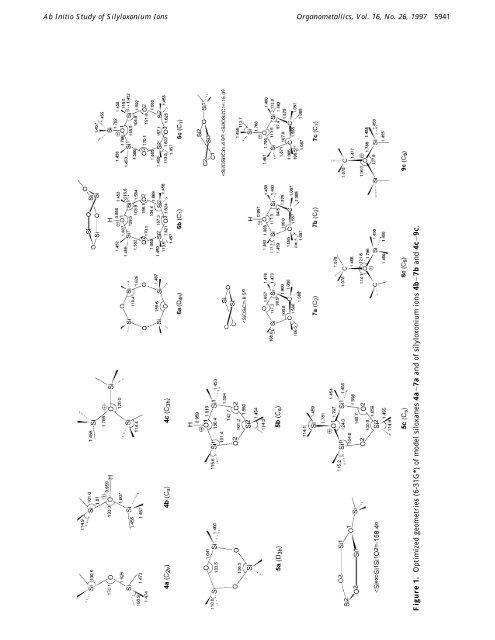
<strong>Ab</strong> <strong>Initio</strong> <strong>Study</strong> <strong>of</strong> <strong>Silyloxonium</strong> <strong>Ions</strong> Organometallics, Vol. 16, No. 26, 1997 5941 Figure 1. Optimized geometries (6-31G*) <strong>of</strong> model siloxanes 4a-7a and <strong>of</strong> silyloxonium ions 4b-7b and 4c-9c.
5942 Organometallics, Vol. 16, No. 26, 1997 Cypryk and Apeloig Table 1. Calculated Geometrical Parameters a <strong>of</strong> the SiOSi Unit in Siloxanes 4a-7a, Protonated Siloxanes 4b-7b, and Silylated Siloxanes 4c-7c species param siloxane (na) na + H + na + SiH3 + H3SiOSiH3 (4a) Si-O1.626 1.807, 1.811 1.786 Si-H 1.474 1.456 1.458 Si-O-Si 170.0 130.3 120.0 H-Si-H 108.9 114.7 114.3 (H2SiO)3 (5a) Si-O1.640 1.819 1.797 Si-H 1.466 1.453 1.454 Si-O-Si 133.5 130.4 124.0 O-Si-O 106.5 101.4 104.8 H-Si-H 110.8 115.6 115.2 (H2SiO)4 (6a) Si-O1.626 1.806 1.785 Si-H 1.467 1.453 1.454 Si-O-Si 159.6 129.5 118.5 O-Si-O 110.4 103.9, 106.7 104.9, 106.2 (CH2SiH2)2O (7a) Si-O 1.657 1.808 1.808 Si-H 1.476 1.459 1.460 Si-O-Si 117.2 117.3 113.5 O-Si-C 99.9 94.5 97.4 H-Si-H 108.6 113.5 112.8 a Bond distances in angstroms and bond angles in degrees. Table 2. GIAO and IGAIM 29 Si NMR Chemical Shifts (Relative to TMS a ) <strong>of</strong> Siloxanes and <strong>Silyloxonium</strong> <strong>Ions</strong> Calculated Using B3LYP/6-311+G**//6-31G* species GIAO IGAIM exptl b 4a -50.9 -45.8 -38.0 (7.0) 4b 17.1 21.7 (22.9) c 4c 11.0 16.3 (51.1) d 5a 5b -41.0 -36.3 (-9.2) Si1 -12.7 -7.9 Si2 5c -29.6 -24.9 Siexo -13.0 -7.9 (50.2) d Si1 -21.1 -15.8 (10.0) Si2 -31.3 -26.5 (-9.6) 6a -60.2 -54.9 -47.2e (-19.4) 8c 29.0 32.6 (66.9) f 9c 20.1 24.4 (59.0) g a δ(TMS) ) 339.5 ppm (GIAO), 343.5 ppm (IGAIM). b Values in parentheses correspond to the permethyl-substituted analogues. 24 c Reference 3. d Reference 5. e Reference 7. f Chemical shift in Me3SiOEt2 + . 4 g Chemical shift in (Me3Si)2OEt + . 4 The calculated 29 Si chemical shifts δ (at B3LYP/6- 311+G**//HF/6-31G*), <strong>of</strong> H3SiOSiH3 are -50.0 ppm (GIAO) and -46.2 ppm (IGAIM). These values are 8-12 ppm higher than the reported experimental vaue <strong>of</strong> -38.0 ppm. 24 However, calculations using the MP2/ 6-31G* optimized geometry gave 29 Si chemical shifts <strong>of</strong> -41.4 ppm (GIAO) and -37.0 ppm (IGAIM), in very good agreement with experiment. This suggests that for correct replacement <strong>of</strong> the magnetic properties <strong>of</strong> siloxanes, the correct Si-O-Si angle should be used. The measured chemical shift <strong>of</strong> Me3SiOSiMe3 is 6.8 ppm, ca. 45 ppm downfield from that <strong>of</strong> 4a. 24 In order to test the reliability <strong>of</strong> the calculated 29 Si chemical shifts <strong>of</strong> 4b,c and <strong>of</strong> 5a-c, we compared them with the well-known experimental chemical shifts <strong>of</strong> their methyl-substituted analogues, assuming that substitution <strong>of</strong> Si-H bySi-Me is additive and is independent <strong>of</strong> the siloxane structure. This assumption can be supported by comparison <strong>of</strong> known chemical shifts <strong>of</strong> linear and cyclic oligosiloxanes with those <strong>of</strong> their methylated analogues. The SiH2 groups in linear and unstrained cyclic siloxanes appear in the range -48 to -52 ppm, Table 3. Calculated Charges (6-31G*) around the Oxonium Center in Model Siloxanes According to Mulliken and NBO Population Analysisa Mulliken NBO a b c a b c 4 H(O) 0.55 0.58 O -0.84 -0.95 -1.02 -1.35 -1.16 -1.32 Si 0.92 0.91 0.91 1.47 1.37 1.36 H(Si) -0.17 -0.06 -0.08 -0.27 -0.19 -0.19 SiH3 b 0.42 0.7 0.67 0.67 0.78 0.78 5 H1 0.54 0.57 Siexo 0.91 1.35 H3Siexo 0.66 0.76 O1 -0.87 -0.96 -1.03 -1.35 -1.18 -1.35 Si1 1.21 1.22 1.22 1.93 1.9 1.89 H(Si1 ) -0.17 -0.09 -0.09 -0.29 -0.24 -0.24 Si1H2 b 0.87 1.04 1.03 1.35 1.42 1.41 6 H1 0.54 0.57 Siexo 0.9 1.36 H3Siexo 0.65 0.64 O1 -0.84 -0.95 -1.02 -1.37 -1.18 -1.34 Si1 1.2 1.21 1.23 1.95 1.89 1.89 H(Si1 ) -0.18 -0.1 -0.09 0.29 0.23 -0.21 Si1H2 b 0.84 1.01 1.0 1.37 1.45 1.49 7 H1 0.55 0.58 Siexo 0.91 1.37 H3Siexo 0.66 0.75 O1 -0.87 -0.95 -1.02 -1.32 -1.16 -1.33 Si2 1.02 1.01 1.02 1.66 1.61 1.61 H(Si2 ) -0.17 -0.08 -0.08 -0.27 -0.21 -0.21 Si2H2 b 0.69 0.86 0.89 1.13 1.2 1.2 a For atom numbering see Chart 1. b Charge on the entire group. whereas SiHMe groups appear at -34 to -37 ppm and SiMe2 groups at -19 to -24 ppm. 7,24 Thus, substitution <strong>of</strong> one H by Me shifts the 29 Si signal by 13-15 ppm downfield. Disiloxanes R3SiOSiR3, where R ) HorMe, obey this correction as well. 24 Further support for this assumption is provided by the fact that the difference between the calculated chemical shift <strong>of</strong> 5a and that measured for D3 is 31.8 ppm (GIAO) and 27.0 ppm (IGAIM), as expected for the substitution <strong>of</strong> two Si-H bonds by two Si-Me groups. For silyloxonium cations the change in the 29 Si chemical shift due to substitution <strong>of</strong> Si-H bySi-Me is difficult to determine because <strong>of</strong> the lack <strong>of</strong> experimental data. For instance, according to IGAIM calculations, the 29 Si signal <strong>of</strong> (H3Si)3O + is shielded by 35 ppm compared to that <strong>of</strong> (Me3Si)3O + . 6 This difference between the 29 Si chemical shifts <strong>of</strong> the exocyclic silicon atoms in 5c and 2 is 58 ppm (IGAIM), while the corresponding difference between the endocyclic Si 1 atoms in these species is 26 ppm. 6 Calculations suggest that in the silylated disiloxane 4c the Si atoms are more shielded than those in the corresponding protonated analogue 4b. This is in contrast to the data reported for (Me3Si)2OH + and (Me3Si)3O + (Table 2). 4,6 Charge Distribution and Orbital Analysis. The atomic charges in all the studied species were calculated using both the Mulliken population analysis 11 and the natural population analysis (NPA), which is based on natural bond orbital (NBO) theory. 12 The results are collected in Table 3, and the numbering system is given in Chart 1. The two methods gave different absolute values (as is well-known) but similar qualitative results, and they are therefore discussed together. The calculations show that the charge at the silicon atoms directly bonded to the oxonium oxygen is approximately the same as in the corresponding neutral
- Page 1 and 2: 5938 Organometallics 1997, 16, 5938
- Page 3: 5940 Organometallics, Vol. 16, No.
- Page 7 and 8: 5944 Organometallics, Vol. 16, No.
- Page 9 and 10: 5946 Organometallics, Vol. 16, No.
- Page 11 and 12: 5948 Organometallics, Vol. 16, No.