Trimetox 6seitig engl - Veyx-Pharma GmbH
Trimetox 6seitig engl - Veyx-Pharma GmbH
Trimetox 6seitig engl - Veyx-Pharma GmbH
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Trimethoprim and sulfamerazine have as monosubstances, in each case a<br />
bacteriostatic effect, but as a combination, a bactericidal one. This is caused by<br />
the double blockade of the bacterial cell metabolism (folic acid synthesis) which<br />
leads to an irreversible damage of the bacteria. Obviously, this double blockade<br />
provides an increased efficiency in the sense of a potentiation and furthermore,<br />
it seems to prevent the danger of the development of resistance to a far extent.<br />
The efficacy of trimethoprim combined with sulfamerazine is synergistic, i.e. the<br />
effect of the combination is considerably stronger than those of the two individual<br />
substances.<br />
This combination has an antibacterial effect against many gram-positive and<br />
gram-negative pathogens (E. coli, species of Shigella, Klebsiella and Pasteurella,<br />
Proteus vulgaris, Staphylococci, Streptococci, Pneumococci, Salmonellae and<br />
others).<br />
<strong>Trimetox</strong>® Powder is easily digested and both foals and mature horses take it in<br />
without problem via the feed.<br />
<strong>Pharma</strong>cokinetic studies with oral application of <strong>Trimetox</strong>® Powder gave a high<br />
bioavailability for the component substances sulfamerazine and trimethoprim.<br />
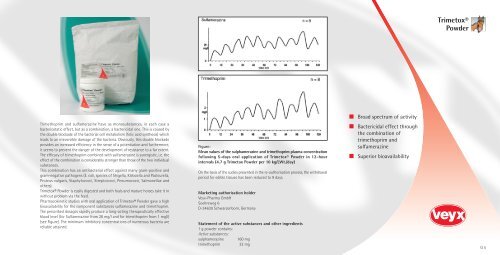
The prescribed dosages rapidly produce a long-acting therapeutically effective<br />
blood level (for Sulfamerazine from 20 mg/l and for trimethoprim from 1 mg/l)<br />
(see Figure). The minimum inhibitory concentrations of numerous bacteria are<br />
reliable attained.<br />
Figure:<br />
Mean values of the sulphamerazine and trimethoprim plasma concentration<br />
following 5-days oral application of <strong>Trimetox</strong>® Powder in 12-hour<br />
intervals (4.7 g <strong>Trimetox</strong> Powder per 10 kg/LWt./day)<br />
On the basis of the sudies presented in the re-authorisation process, the withdrawal<br />
period for edible tissues has been reduced to 9 days.<br />
Marketing authorisation holder<br />
<strong>Veyx</strong>-<strong>Pharma</strong> <strong>GmbH</strong><br />
Soehreweg 6<br />
D-34639 Schwarzenborn, Germany<br />
Statement of the active substances and other ingredients<br />
1 g powder contains:<br />
Active substances:<br />
sulphamerazine 160 mg<br />
trimethoprim 32 mg<br />
� Broad spectrum of activity<br />
� Bactericidal effect through<br />
the combination of<br />
trimethoprim and<br />
sulfamerazine<br />
� Superior bioavailability<br />
<strong>Trimetox</strong>®<br />
Powder<br />
G 5
Excipient:<br />
Glucose-monohydrate<br />
<strong>Pharma</strong>ceutical form and content<br />
Powder (500 g, 1,500 g, 5,000 g)<br />
Substance or indication group<br />
Sulfonamide-trimethoprim-combination<br />
Indications<br />
For the treatment of the following diseases caused by sulfamerazine and<br />
trimethoprim sensitive pathogens during the early stages of the infection:<br />
- bacterial infections of the upper respiratory tract<br />
- infections of the gastrointestinal tract<br />
- infections of the urogenital tract<br />
The administration of <strong>Trimetox</strong>® Powder should be carried out based on an<br />
antibiogram.<br />
Contraindications<br />
- severe liver and kidney function disorders<br />
- damage to the haematopoietic system<br />
- hypersensitivity to sulfonamides or trimethoprim<br />
- resistance to sulfonamides or trimethoprim.<br />
- diseases relating to a severe reduction in fluid intake and severe losses of fluids<br />
Adverse reactions<br />
After the application of <strong>Trimetox</strong>® Powder the following can occur:<br />
- Liver damages<br />
- Kidney damages, along with haematuria, crystalluria, kidney colic, lack of<br />
appetite, laboured urination<br />
- Allergic reactions<br />
- Changes in the blood profile<br />
- Digestive disorders<br />
In case any of the side-effects given above occurring, then the <strong>Trimetox</strong>® Powder<br />
must be withdrawn immediately.<br />
Counter measures<br />
In case of indications of kidney damage: fluid intake and alkalising of the urine.<br />
In case of anaphylactic shock: epinephrine (adrenalin) and glucocorticoids i.v.<br />
In case of allergic skin reactions, antihistamines and/or glucocorticoids.<br />
If you notice any serious or other effects not mentioned in this leaflet, please<br />
inform your veterinary surgeon or pharmacist.<br />
Target species<br />
Horses<br />
Dosage for each species, routes and method of administration<br />
To be given orally via the feed.<br />
For the treatment of individual animals.<br />
The following dosage rates are based on the quantity of the active substance for<br />
sulfamerazine and trimethoprim mixed at a fixed ratio of 5 + 1.<br />
90 mg total active substance/kg body weight/day equates to<br />
4.7 g <strong>Trimetox</strong>® Powder per 10 kg body weight/day<br />
The daily dose should be split into two equal a.m. and p.m. doses.<br />
The treatment is carried out over 3 to 5 consecutive days and for 2 days,<br />
respectively, after the disease symptoms disappear (in case of Salmonella 5 days).<br />
In case of chronic diseases of the respiratory tract and lungs in horses, the<br />
treatment duration of 5 to 10 days may be indicated.<br />
Prior to each application the powder should be freshly mixed into a portion of<br />
feed, ensuring a complete thorough mix is achieved. It should be fed prior to the<br />
main feed ration.<br />
Where no visible improvement in the disease situation is noted following 3<br />
treatment days, then the diagnosis needs to be reassessed and where appropriate<br />
a change in therapy instituted. Confirmation of the pathogen's susceptibility is<br />
recommended.<br />
Advice on correct administration<br />
Use in gestating animals and neonates' demands a strict adherence to the<br />
indication.<br />
To avoid sensitization or contact dermatitis, direct contact with the skin should<br />
be avoided along with inhalation during use and administration. In this instance<br />
suitable gloves and a mask should be worn.<br />
Withdrawal period<br />
Horse: edible tissues 9 days<br />
Should not be administered to mares if their milk is intended for human<br />
consumption.<br />
Special storage precautions<br />
Do not use after the expiry date stated on the label and the outer carton.<br />
Do not store above 25 °C.<br />
The powder must be protected from light.<br />
Special warnings<br />
Interaction with other medicaments and other forms of interaction:<br />
<strong>Trimetox</strong>® Powder should not be used at the same time as phenylbutazone.<br />
Mixing with other medicaments should be avoided because of possible<br />
incompatibilities.<br />
Overdose:<br />
Following an overdose atactic movements, muscle twitching and convulsions,<br />
along with comatose conditions and liver damages can occur. <strong>Trimetox</strong>® Powder<br />
must be discontinued immediately. The neurotropic effects are to be treated<br />
symptomatically through the giving of central nervous system sedating substances<br />
(e.g. barbiturates).<br />
In addition to the giving of vitamin K or folic acid, an increase in the renal<br />
excretion of sulfonamide is prescribed through the giving of alkalising substances<br />
(e.g. sodium bicarbonate).<br />
Special precautions for the disposal of unused product or waste materials,<br />
if any:<br />
Any unused veterinary medicinal product or waste materials derived from such<br />
veterinary medicinal product should be disposed of in accordance with local<br />
requirements.<br />
<strong>Veyx</strong>-<strong>Pharma</strong> <strong>GmbH</strong> · Soehreweg 6 · 34639 Schwarzenborn · Germany<br />
Phone 0049 5686 9986-0 · Fax 0049 5686 1489 · E-Mail zentrale@veyx.de<br />
www.veyx.de<br />
Stand 03/2007