Substitution von molekularen Klammern an den Naphthalin ...
Substitution von molekularen Klammern an den Naphthalin ...
Substitution von molekularen Klammern an den Naphthalin ...
Erfolgreiche ePaper selbst erstellen
Machen Sie aus Ihren PDF Publikationen ein blätterbares Flipbook mit unserer einzigartigen Google optimierten e-Paper Software.
Experimenteller Teil<br />
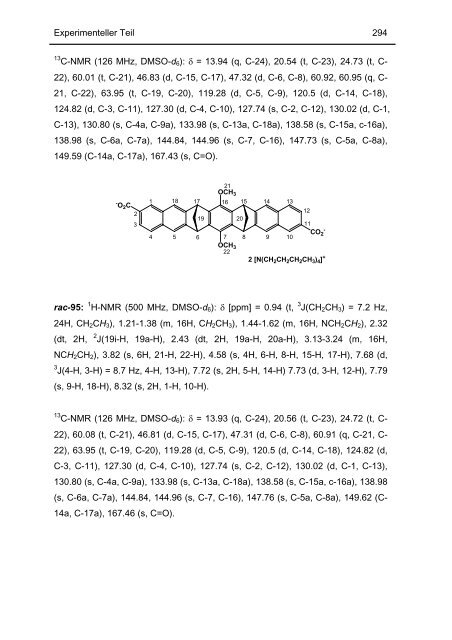
13 C-NMR (126 MHz, DMSO-d6): δ = 13.94 (q, C-24), 20.54 (t, C-23), 24.73 (t, C-<br />
22), 60.01 (t, C-21), 46.83 (d, C-15, C-17), 47.32 (d, C-6, C-8), 60.92, 60.95 (q, C-<br />
21, C-22), 63.95 (t, C-19, C-20), 119.28 (d, C-5, C-9), 120.5 (d, C-14, C-18),<br />
124.82 (d, C-3, C-11), 127.30 (d, C-4, C-10), 127.74 (s, C-2, C-12), 130.02 (d, C-1,<br />
C-13), 130.80 (s, C-4a, C-9a), 133.98 (s, C-13a, C-18a), 138.58 (s, C-15a, c-16a),<br />
138.98 (s, C-6a, C-7a), 144.84, 144.96 (s, C-7, C-16), 147.73 (s, C-5a, C-8a),<br />
149.59 (C-14a, C-17a), 167.43 (s, C=O).<br />
-O2C 2<br />
3<br />
1<br />
18<br />
17<br />
21<br />
OCH3 16 15<br />
19 20<br />
4 5 6 7<br />
OCH3 22<br />
8 9 10<br />
14<br />
13<br />
12<br />
11<br />
- CO2 2 [N(CH 2CH 2CH 2CH 3) 4] +<br />
rac-95: 1 H-NMR (500 MHz, DMSO-d6): δ [ppm] = 0.94 (t, 3 J(CH2CH3) = 7.2 Hz,<br />
24H, CH2CH3), 1.21-1.38 (m, 16H, CH2CH3), 1.44-1.62 (m, 16H, NCH2CH2), 2.32<br />
(dt, 2H, 2 J(19i-H, 19a-H), 2.43 (dt, 2H, 19a-H, 20a-H), 3.13-3.24 (m, 16H,<br />
NCH2CH2), 3.82 (s, 6H, 21-H, 22-H), 4.58 (s, 4H, 6-H, 8-H, 15-H, 17-H), 7.68 (d,<br />
3 J(4-H, 3-H) = 8.7 Hz, 4-H, 13-H), 7.72 (s, 2H, 5-H, 14-H) 7.73 (d, 3-H, 12-H), 7.79<br />
(s, 9-H, 18-H), 8.32 (s, 2H, 1-H, 10-H).<br />
13 C-NMR (126 MHz, DMSO-d6): δ = 13.93 (q, C-24), 20.56 (t, C-23), 24.72 (t, C-<br />
22), 60.08 (t, C-21), 46.81 (d, C-15, C-17), 47.31 (d, C-6, C-8), 60.91 (q, C-21, C-<br />
22), 63.95 (t, C-19, C-20), 119.28 (d, C-5, C-9), 120.5 (d, C-14, C-18), 124.82 (d,<br />
C-3, C-11), 127.30 (d, C-4, C-10), 127.74 (s, C-2, C-12), 130.02 (d, C-1, C-13),<br />
130.80 (s, C-4a, C-9a), 133.98 (s, C-13a, C-18a), 138.58 (s, C-15a, c-16a), 138.98<br />
(s, C-6a, C-7a), 144.84, 144.96 (s, C-7, C-16), 147.76 (s, C-5a, C-8a), 149.62 (C-<br />
14a, C-17a), 167.46 (s, C=O).<br />
294